 
When a gas is put under pressure by reducing the volume of the container, the space between particles is reduced and the gas is compressed, according to NASA's Glenn Research Center. If unconfined, the particles of a gas will spread out indefinitely if confined, the gas will expand to fill its container. In a gas, the particles have a great deal of space between them and have high kinetic energy. Much like solids, liquids (most of which have a lower density than solids) are incredibly difficult to compress. Therefore, the liquid will conform to the shape of its container. In a liquid, the particles are more loosely packed than in a solid and are able to flow around each other, giving the liquid an indefinite shape. 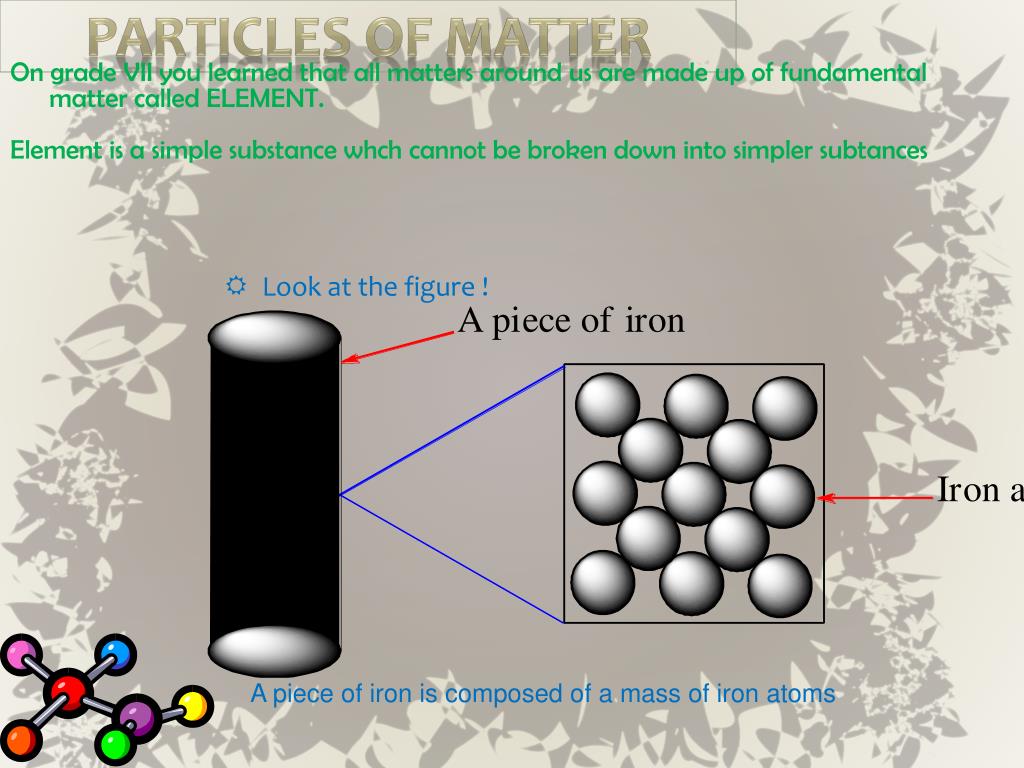
Solids also have a high density, meaning that the particles are tightly packed together. 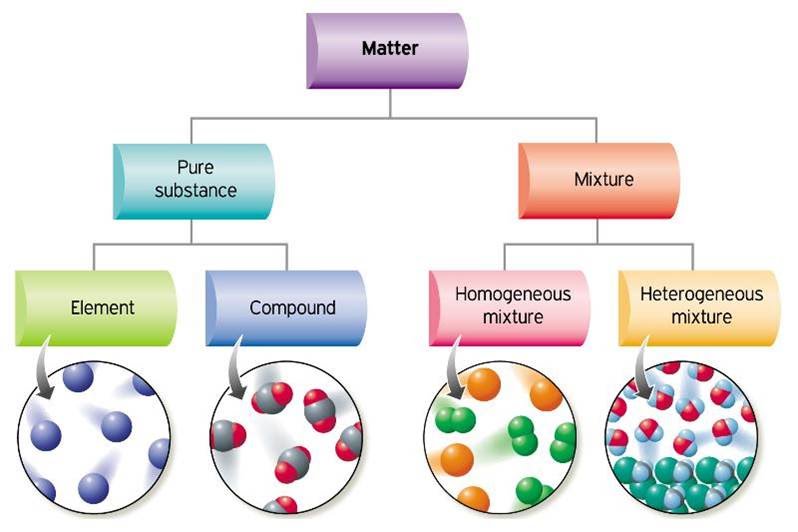
Solids have a definite shape, as well as mass and volume, and do not conform to the shape of the container in which they are placed. Because of this, particles in a solid have very low kinetic energy. The electrons of each atom are constantly in motion, so the atoms have a small vibration, but they are fixed in their position. In a solid, particles are packed tightly together so they don't move much. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
